 
This now gives N and all of the O a share in 8Įlectrons, the nitrogen and oxygen are joined by a double bond, which would be represented as N=O. Make a second bonding pair between the oxygen and nitrogen. 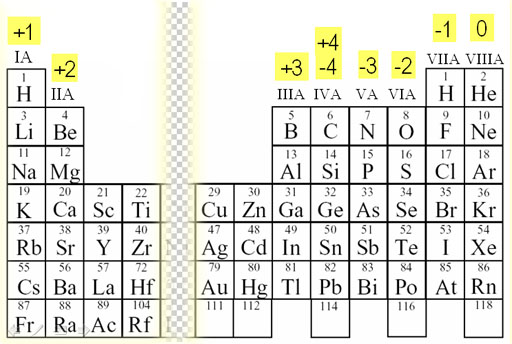
One of the lone pairs of electrons from an oxygen, and To give nitrogen a chare in 8 electrons, move However, nitrogen only has a share in 6 electrons. Surrounded by 8electrons and the hydrogen has 2 electrons. H + in aqueous solutions, is always bonded to an Nitric acid is one of the " oxy-acids", like sulfuric, phosphoric and choric The edge of a molecule, since it can only form one bond. Total electrons = 5 + (3圆) + 1 = 24 electrons What is the Lewis Structure for nitric acid, HNO 3? There are 3 bonding pairs of electrons and 10 lone pairs of electrons. Total electrons = 5 + (3x7) = 26 electronsĮlectrons around each of the fluorine atoms.Īnd gives every atom in the molecule a share in 8 valence electrons, an octet. What is the structure of nitrogen trifluoride, NF 3? Met for the central atom, move lone pairs of electrons to make multiple bonds Place all remaining electrons on the central atom. Using pair of electrons, make bonds between the central atom and each For polyatomic cations, remove enough electrons toĮlement in the center of the molecule (never hydrogen).ģ. For polyatomic anions, add enough extra electrons to account for the negativeĬharge. Examine the atoms in the molecule and determine how many valence electrons they each contribute. Of steps that can be followed to simplify the determination of a Lewisġ. Hydrogen, which fills its valence shell (1 s) There are exceptions to this rule, which we Of valence electrons, to attain a noble gas configuration. It is assumed that most elements require an octet Of valence electrons in the formation of chemical bonds. Lewis structures are used to help visualizeĢ-dimansional representations of molecular structures, based on the arrangement 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
